
Clinical Reports · Klinische Mitteilungen · Communications cliniques
Withdrawal Reactions from Chlordiazepoxide ("Librium")
Leo E. Hollister, Francis P. Motzenbecker & Roger O. Dean
(Received August 29, 1960)
Psychopharmacologia 2, 63-68 (1961)
Veterans Administration Hospital, Palo Alto, California
With 1 Figure in the Text
Chlordiazepoxide is a newly introduced tranquilizer chemically unrelated to other groups of psychotherapeutic drugs. Four pharmacologic effects have been described: 1. tranquilization, 2. muscle relaxation, 3. anticonvulsant activity, and 4. appetite stimulation (RANDALL 1960). Preliminary clinical trials, primarily in patients with psychoneurosis or emotional upsets associated with somatic illnesses, have been uniformly encouraging (TOBIN 1960; KINROSS-WRIGHT 1960; ENGLISH 1960; HARRIS 1960). Thus far, habituation has not occurred in patients under treatment with usual doses at 30 to 75 mg daily. Larger doses were used in this study, both to determine therapeutic efficacy in more severely ill patients and possible withdrawal reactions.
Preliminary study
Thirty-six hospitalized psychiatric patients were treated with 100 to 600 mg of chlordiazepoxide daily for one to seven months (most more than three months). Their median age was 35 years, but in each instance, illnesses were chronic. Frequent laboratory and clinical observations were made during this study. Nine of the 36 patients experienced clinically significant improvement: 4 of 10 with anxiety reactions, 5 of 22 with schizophrenic reactions, none of 4 with other diagnoses. Dizziness, weakness, unsteadiness and sleepiness were common side-effects, usually from too rapid in crease from the initial dosage of 25 mg three times daily. Three patients became agitated from high doses (300 mg or more), requiring reduction. Twenty-two had one or more sporadic laboratory abnormalities: leucocytosis (total count over 13,500 per cu ml) in 18, eosinophilia (more than 7 percent) in 1, elevation of serum glutamic oxalacetic transaminase (SGO-T) titer in 4. Only 1 of the elevated SGO-T titers remained on rechecking, and none were associated with other abnormal hepatic tests.
Withdrawal study
Eleven patients were abruptly withdrawn by switching to placebo without their knowledge, though with the knowledge of the observers Doses when withdrawn ranged from 300 to 600 mg daily, 6 patient; receiving the latter dose. Duration of treatment varied from two to six months, 10 patients having been treated for five months or longer.
For several days prior to the switch to placebos, patients were observed carefully for symptoms and signs ordinarily associated with withdrawal reactions, such as insomnia, agitation, depression, worsening of psychosis, tremor, twitches, seizures, nausea, vomiting, loss of appetite cyanosis, sweating, or other new symptoms. If present, these were recorded on a symptom-sign check-list. Records of blood pressure and pulse rate were also made, all observations being continued for a ten. day period following switch to placebos. Immediately after the last dose of drug, blood was drawn for determinations of plasma levels of chlordiazepoxide, and an electroencephalogram was obtained. Plasma levels of drug were determined on each of the first four days following withdrawal and on the seventh day in some patients. Electroencephalograms were repeated at twenty-four and seventy-two hours following withdrawal from the drug.
Ten of the 11 patients experienced new symptoms or signs following drug withdrawal (Table). Six patients became depressed after being switched to placebos. Psychoses were aggravated in 5, insomnia and agitation appeared in 5, 4 had loss of appetite but only 1 was nauseated Two patients had major convulsions on the seventh and on the eighth day following drug withdrawal. One patient had a single previous seizure associated with earlier drug therapy; the other had no knowr seizures before. Another patient, not in the formal withdrawal study; experienced two new major seizures twelve days after abrupt withdrawal from a daily dose of 300 mg. He had been treated with chlordiazepoxide for three months but had become agitated at the higher dose range. One patient developed twitching and excessive perspiration on the eighth day after withdrawal. In general, symptoms appeared from two to nine days after withdrawal, most appearing between the fourth and eighth days. By the tenth day, symptoms were either waning in intensity or had subsided. Symptoms from withdrawal of chlordiazepoxide were slower to develop and less acute than those following withdrawal of barbiturates or meprobamate. Nevertheless, they were definite and consistent with a withdrawal reaction.
Table. Clinical and electroencephalographic findings in eleven patients withdrawn from large doses of chlordiazepoxide
| Patient | Age/Sex | Diag. | Duration Treatment |
Daily dose (mg) |
EEG Tracings | Withdrawal signs or symptoms |
||
|---|---|---|---|---|---|---|---|---|
| Control | 24 hrs. | 72 hrs. | ||||||
| Da. | 37/M | Anx. | 6 mos. | 400 | Mild Abn. 10/s |
N | N | Depression, 7-8th day |
| Hu. | 49/M | Schiz. | 5 mos. | 600 | N | Borderline mixed |
N | Seizure, 8th day |
| Kl. | 28/M | Schiz. | 5 mos. | 600 | Mild Abn. 7/s and 20-25/s |
Mild Abn. 8-22/s |
Insomnia, 4-8th day. Psychosis worse, 2-9th day | |
| Md. | 31/M | Schiz. | 5 mos. | 400 | N | Mild Abn. disorg. |
Mild Abn. disorg. |
Psychosis worse, 4-7th day. Loss appetite, 4-6th day |
| Ri. | 35/M | Schiz. | 5 mos. | 450 | Mild Abn. 6-7/s |
Mild Abn. 7-8/s |
N | Depression, 7-9th day |
| Sh. | 24/M | Pers. Dis. |
6 mos. |
600 | Mild Abn. Fast |
Mild Abn. fast |
N | Agitation, 2-6th day. Nausea, 3-6th day. Loss appetite, 4-8th day. Seizures, 7th day. Depression, 8-9th day |
| Wi. | 29/M | Schiz. | 6 mos. | 600 | Mild Abn. 6-8/s |
Mild Abn. 6-9/s |
Mild Abn. slow |
Agitation, 1-9th day. Depression, 2-8th day. Loss appetite, 6th day. Psychosis worse, 7-9th day. Twitching, sweating, 9th day |
| Fa. | 40/M | Schiz. | 5 mos. | 600 | N | Mild Abn. 7-8/s |
N | Depression, 2-9th day. Psychosis worse, 3-9th day |
| Ga. | 32/M | Schiz. | 6 mos. | 300 | N | N | N | Depression, 2-8th day. Loss appetite, 2-8th day. Psychosis worse, 3-8th day. Agitation, 4-6th day |
| Go. | 34/M | Schiz. | 5 mos. | 400 | N | N | N | Insomnia, 3-7th day. Agitation, 2-4th day |
| Ro. | 48/M | Anx. | 2 mos. | 600 | N | N | N | None |
When withdrawn from chlordiazepoxide, 6 patients had normal electroencephalographic tracings. The remaining 5 showed only mild abnormalities, consisting of slow activity (6-10/s) in 3, mixed fast (20-25/s) and slow (7/s) in 1; and fast (20-25/s) activity in 1. At the end of twenty-four hours, 3 previously normal tracings had become abnormal, 1 showing borderline abnormal mixed fast and slow activity, 1 showing disorganized alpha rhythm, and 1 showing paroxysmal slowing (7-8/s). At the end of seventy-two hours, 2 of the patients with newly-appearing abnormal tracings had returned to the normal pattern, 1 continuing to show mild abnormalities due to disorganization of the alpha rhythm. Of the 5 patients with mildly abnormal tracings at the time of drug withdrawal,1 developed a normal tracing at the end of twenty four hours and retained this at the end of seventy two hours. Two other patients had normal tracings at the end of seventy two hours though retaining their abnormalities during the twenty-four-hour period. Two patients retained the original abnormalities for the entire seventy-two-hour period.
Both patients later developing grand mal seizures had normal tracings seventy-two hours after drug withdrawal. Tracings taken following the seizures showed a mildly abnormal slow (8/s) tracing in 1 and a moderately abnormal slow (6/s) tracing in the other.
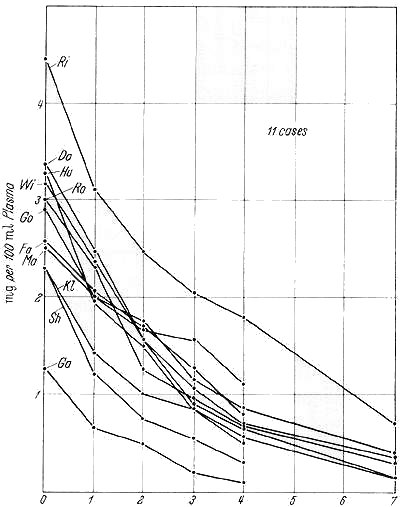
Fig. 1. Change in plasma levels of chlordiazepoxide after abrupt discontinuation of large chronic doses |
Changes in plasma levels of chlordiazepoxide following abrupt discontinuation of large chronic doses are shown in Fig. 1. At the end of one day, the mean percentage of the original level of the drug remaining was 68 percent; at the end of two days, 50 percent; three days, 36 percent; four days, 25 percent; and seven days, 10 percent. The appearance of seizures in 2 patients apparently coincided with nearly-complete disappearance of drug from the plasma. Discussion Therapeutic results were not outstanding in this group of patients because of their chronicity and the fact that many were schizophrenics, for whom chlordiazepoxide is not usually considered to be the drug of choice. Even at the monumental doses used, side effects were few. Sporadically occurring laboratory abnormalities are often found when repetitive tests are performed in hospitalized psychiatric patients and do not necessarily indicate toxicity (HOLLISTER et al. 1960a). Although differentiating withdrawal reactions from recrudescent psychiatric symptoms after drug withdrawal is always difficult, a number of factors favored the former interpretation of the newly-appearing symptoms and signs. First, the frequency (10 of 11 patients) of new symptoms or signs soon after withdrawal. Second, their coincidence with decreasing plasma levels of drug. Third, postwithdrawal seizures in 2 patients. Fourth, a slower onset and subtler development of this syndrome than that from meprobamate or barbiturates. This difference is consonant with the slower decline in plasma levels of chlordiazepoxide, whose half-life is forty-eight hours as compared with twenty-four hours for meprobamate (HOLLISTER and GLAZENER 1960 b). Chronic administration of large doses of chlordiazepoxide produced mild electroencephalographic abnormalities, mainly slowing. Few additional changes were evoked during the first seventy-two hours after drug withdrawal, possibly due to the slow disappearance of the drug from plasma. Routine tracings taken seven to eight days following withdrawal might have detected more abnormalities, as 2 patients developed frank seizures then. |
Summary
Ten of 11 patients abruptly switched to placebos after several months of high daily doses (300-600 mg, 8-20 times the usual therapeutic range) of chlordiazepoxide experienced new symptoms and signs soon after. Depression, aggravation of psychoses, agitation, insomnia, loss of appetite, and nausea appeared between two and eight days following abrupt withdrawal. Two patients had seizures at seven and eight days. A third patient, not in the withdrawal study, had a seizure twelve days after discontinuation of a 300 mg daily dose. Plasma levels of drug indicated a half-life of approximately forty-eight hours following discontinuation of chronic high doses. Minor electroencephalographic abnormalities (mainly slowing) were produced by chronic administration of these exceedingly high doses. If large doses of chlordiazepoxide are used, the drug should not be abruptly discontinued.
Acknowledgement. This study was supported by grant MY 3030, National Institute of Mental Health. Mrs. FRANCES MOORE performed the biochemical studies. Supplies of chlordiazepoxide were given by Roche Laboratories, Nutley, N. J.
References
ENGLISH, D.C.: Librium a new nonsedative neuroleptic drug: A clinical evaluation Curr. Ther. Res. 2 (3), 88-91 (1960).
HARRIS, T. H.: Methaminodiazepoxide. J. Amer. med. Ass. 172, 1162-1163 (1960).
HOLLISTER, L. E., E. M. CAFFEY Jr. and C. J. KLETT: Abnormal symptoms, signs and laboratory tests during treatment with phenothiazine derivatives. A controlled study. Clin. Pharm. and Ther. 1, 284 (1969a).
HOLLISTER, L. E., and F. S. GLAZENER: Withdrawal reactions from meprobamate, alone and combined with promazine: A controlled study. Psychopharmacologia 1, 336-341 (1960b).
KINROSS-WRIGHT, J., I. M. COHEN and J. A. KNIGHT: The management of neurotic and psychotic states with Ro 5-0690 (Librium). Dis. nerv. Syst. 21, Suppl. 3, 23-26 (1960).
RANDALL, L. D.: Pharmacology of methaminodiazepoxide. Dis. nerv. Syst. 21, Suppl. 3, 7-10 (1960).
TOBIN, J. M., I. F. BIRD and D. E. BOYLE: Preliminary evaluation of Librium (Ro 5-0690) in the treatment of anxiety reactions. Dis. nerv. Syst. 21, Suppl 3, 11-19 (1960).
LEO E. HOLLISTER, M. D.
FRANCIS P. MOTZENBECKER, M. D., ROGER 0. DEGAN, M. D.
Veterans Administration Hospital, Palo Alto, California, U.S.A.
« back · top · www.benzo.org.uk »
